The Dry Cell was discovered by French Scientist G. Leclanche in 1868. It is the most common cell which is the improved version of Leclanche Cell. It is a portable cell and free from liquid. This is why, it is called dry cell as it do not contain any liquid. Dry Cell is widely used in our day to day life in torch, clock, toys etc.
Construction of Dry Cell
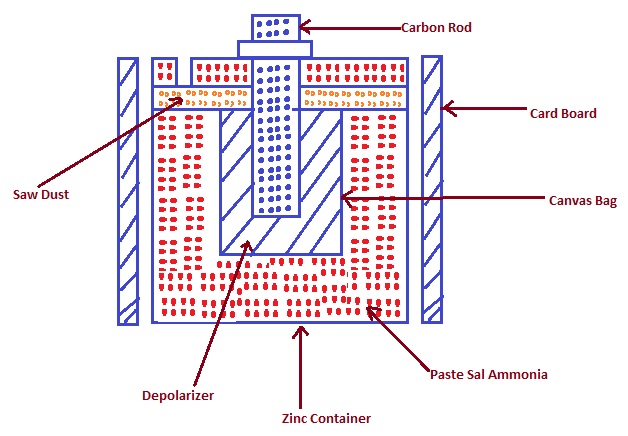
The construction of dry cell is shown in figure below. As can be seen from the figure, Zinc Container acts as cathode i.e. negative electrode while carbon rod acts as positive electrode or anode.
Carbon rod is placed in the middle of Zinc container. It shall be noted that carbon rod is covered with crushed coke and Manganese dioxide and kept in canvas bag. Here the mixture of coke and Manganese dioxide acts as depolarizer. Depolarizer is basically a substance used in a cell to prevent build-up of hydrogen gas bubbles. A battery depolarizer takes up electrons during discharge of the cell and hence depolarizer is always an oxidizing agent.
In Dry Cell, electrolyte is paste of sal ammoniac and Zinc Chloride. Sal Amminiac is a mineral composed of Ammonium Chloride NH4Cl. Electrolyte is filled in between the positive plate i.e. carbon rod and negative plate which is zinc container. Zinc chloride is added in sal ammoniac paste to keep the paste wet. Upper portion of cell is covered with saw dust and pitch compound and a small hole is kept intentionally for venting of cell.
Working Principle of Dry Cell
When dry cell is externally connected with wire, following reactions take place,
Zn + 2NH4 Cl = ZnCl2 + 2NH3 + H2
Here NH3 and H2 are in the form of ions.
Ammonia dissolves in water and after saturation it comes out. It shall be noted here that, H2 reaches to anode i.e. toward the carbon rod and there it reacts with MnO2 to makes water.
2MnO2 + H2 = Mn2O3 + H2O
Normally the emf of dry cell is 1.5 V and its internal resistance lies in between 0.1 to 0.5 ohm. These cells are used in portable equipment like Transistor, Torch, TV remote, remote bell push etc.
I have read many articles on this topic but all of them haven't mentioned about use of MnO2 in cell. Your content is too good. Keep it up. Have a good day 👍
This website has a very good content.
Really very helpful. Keep it up
Thank you Prerna!